News
-

Clinical trials and innovative oncological drugs: does the sponsor influence the selectivity of a clinical trial?
Clinical trials of anticancer drugs are often criticized for their overly selective inclusion criteria. As we know, a…
Leggi » -

"Expert patient": the 110 Cnamc associations ask for the withdrawal of the resolution 702/2022 of the Tuscany Region
“We risk the elimination of participatory practices. The patient is an expert as such and his point of view is…
Leggi » -

Banner of animal rights activists in front of Novartis in Origgio: "Get the beagles out of the laboratories"
Nella serata di giovedì 30 giugno militanti di “Centopercentoanimalisti” si sono recati davanti all’entrata della sede di Origgio della multinazionale…
Leggi » -

Private equity platform TPG invests in leading generic drug maker: DOC Generici
TPG investe nel principale produttore di farmaci generici: DOC Generici L’investimento contribuirà a rafforzare ulteriormente la leadership di DOC Generici…
Leggi » -
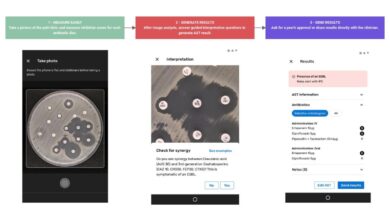
MSF presents the “Antibiogo” App. It will allow non-experts to measure and interpret susceptibility readings.
Antibiogo is a diagnostic aid medical device that aims to help doctors prescribe the most effective antibiotics…
Leggi » -

EFPIA (Federation of European Pharmaceutical Industries). Report 2022: growing sector
Secondo il Rapporto EFPIA 2022 il settore farmaceutico europeo ad oggi rappresenta il 23,4% del mercato mondiale, con 840 mila addetti…
Leggi » -

Critical drugs in Europe. EMA activates i-SPOC, single point of contact for the sector which will inform EMA
I titolari dell’autorizzazione all’immissione in commercio (AIC) possono ora registrare il loro , (i-SPOC) della fornitura e della disponibilità…
Leggi » -

Document from the MINDS for ONE HEALTH Expert Group
We report the position taken by the Minds for One Health (M4OH) group, made up of about sixty experts from different…
Leggi » -

The purchase of drugs in the Cnn class: critical issues and addresses at a legal level
In 2012, with the law n. 189, also known as the Balduzzi Decree, the legislator intended to provide a new instrument…
Leggi » -

AIFA. Launch of the "TrovaNormeFarmaco" portal
L’Agenzia Italiana del Farmaco ha attivato il portale TrovaNormeFarmaco con l’obiettivo di favorire la ricerca e la consultazione della normativa…
Leggi »
